Rapid Molecular Identification of Nontuberculous Mycobacteria (NTM)
Powered by Probe-Based Melting Curve Analysis™ technology for high-multiplex, species-level detection in a single reaction.
Key Highlights
< 3-hour turnaround time
Species-level identification
High-multiplex single-tube detection
Automated interpretation
Clinical Need
The Clinical Challenge of NTM Infections
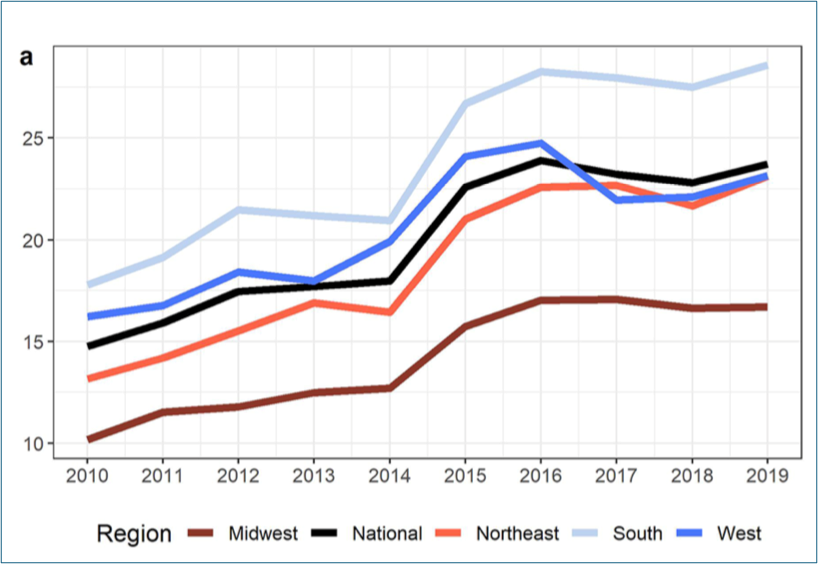
Annual incidence time series per 100k Medicare beneficiaries colored by US region (Midwest, Northeast, South, and West) compared to the national.
(Bents, S.J., Mercaldo, R.A., Powell, C. et al. Nontuberculous mycobacterial pulmonary disease (NTM PD) incidence trends in the United States, 2010–2019. BMC Infect Dis 24, 1094 (2024)).
Nontuberculous mycobacteria (NTM) are increasingly recognized as significant human pathogens, particularly in patients with chronic lung disease or immunocompromised conditions.
Unlike Mycobacterium tuberculosis, NTM comprise a diverse group of species with heterogeneous pathogenicity, transmission patterns, and treatment responses.
Highlights the importance of NTM Species identification
“Correct identification of NTM is important, as it can predict the clinical relevance of an isolate as well as aid in the selection of a treatment regimen. Both molecular and mass spectrometry-based methods can be applied. Molecular identification is the preferred method and can be achieved using probes or gene sequencing.”
Charles L Daley, et al. Treatment of Nontuberculous Mycobacterial Pulmonary Disease: An Official ATS/ERS/ESCMID/IDSA Clinical Practice Guideline, Clinical Infectious Diseases, Volume 71, Issue 4, 15 August 2020, Pages e1–e36,
Clinical challenges:
The treatment is highly species-specific. However, current workflows rely on culture followed by MALDI-TOF or sequencing, which typically takes 2 to 6 weeks.
During this period, clinicians often have to make empiric treatment decisions without species-level information, which can lead to suboptimal therapy and prolonged disease progression.
So there is a clear need for a rapid, species-level diagnostic that can be implemented in routine clinical microbiology labs.
Why NTM Identification is Critical
Treatment regimens are species-dependent and prolonged
Misidentification may lead to ineffective therapy or overtreatment
Culture-based identification can take weeks
Rapid molecular identification is essential for early clinical decision-making
Technology Platform
Probe-Based Melting Curve Analysis™: High-Multiplex Molecular Detection
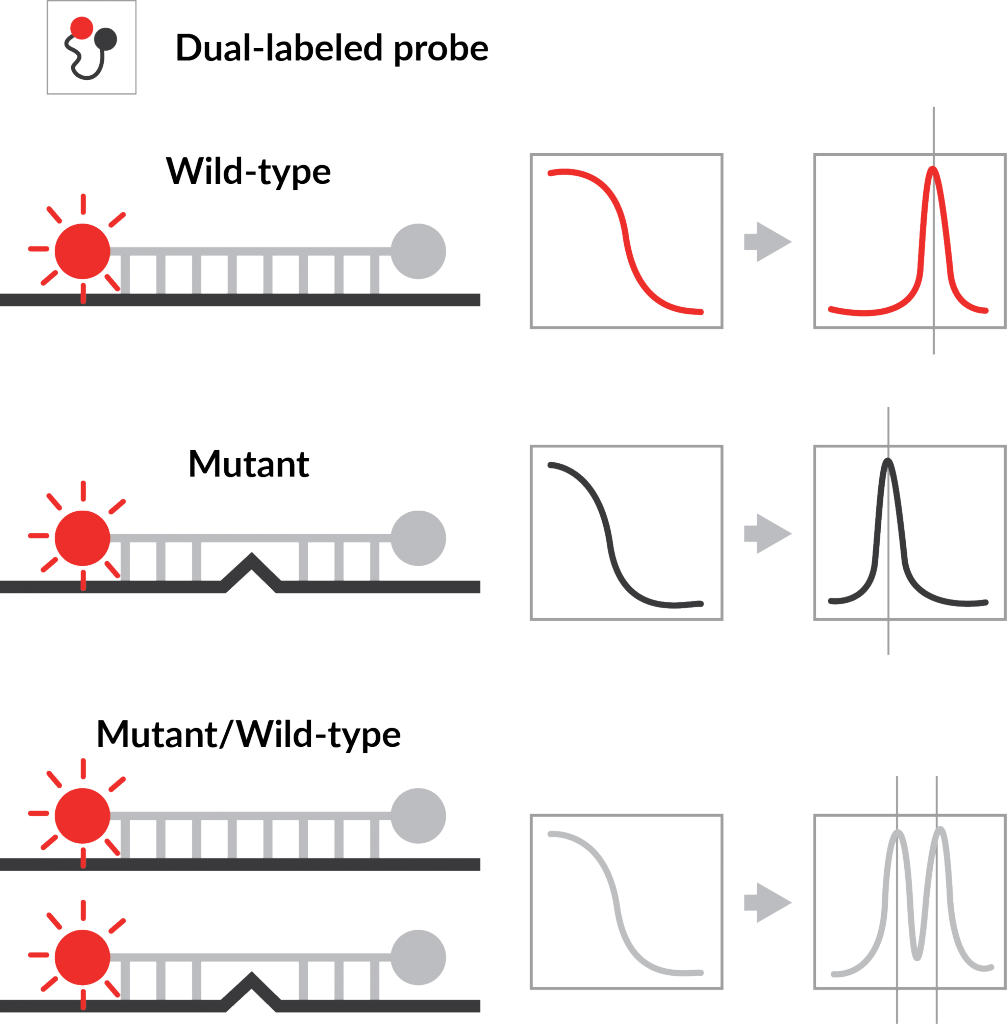
Huang, Q., et al.(2022). Highly multiplex PCR assays by coupling the 5′-flap endonuclease activity of Taq DNA polymerase and molecular beacon reporters. Proceedings of the National Academy of Sciences, 119(9).
The NTM assay is built on the proprietary Probe-Based Melting Curve Analysis platform, which enables high-level multiplexing in a single PCR reaction using melting-temperature–encoded fluorescence signatures.
Each target organism is identified by its unique melting temperature profile, enabling simultaneous detection of multiple species in a single reaction.
Enabling High-Density Multiplexing
Unlike traditional PCR assays limited by fluorescence channels, MeltArray™ significantly expands multiplexing capacity by:
Encoding targets through melting temperature differentiation
Allowing multiple targets per fluorescence channel (up to 12 targets per channel)
Supporting high-throughput detection without increased workflow complexity
This architecture enables broad NTM coverage in a streamlined format.
Product Overview
Comprehensive NTM Identification Panel
Identifying 19 clinically relevant mycobacteria (including M. tuberculosis complex, M. chelonae, M. scrofulaceum, M. bovis, M. smegmatis, M. abscessus, M. kansasii, M. gordonae, M. lentiflavum, M. simiae, M. nonchromogenicum, M. terrae, M. marinum/M. ulcerans, M. fortuitum, M. avium, M. intracellulare, M. xenopi, M. malmoense and M. szulgai) from sputum samples.
The assay is designed to cover the major pulmonary NTM species highlighted in international clinical guidelines, including MAC-associated species, M. abscessus, M. kansasii, and M. xenopi, as well as other clinically encountered mycobacteria, including select non-NTM mycobacteria for differential identification.
The panel supports identification from respiratory specimens such as sputum, as well as cultured isolates.
Workflow & System Integration
Designed for Clinical Laboratory Efficiency
The MeltArray™ NTM solution is optimized for streamlined workflows by either approach:
1. End-to-End Automated Workflow (Sanity™ Platform) Sample → Extraction → Amplification → Detection → Result
• Minimal hands-on time
• Reduced operator variability
• Integrated data analysis2. Flexible PCR-Based Workflow
• Compatible with real-time PCR systems
• Easy integration into existing lab infrastructure
Performance
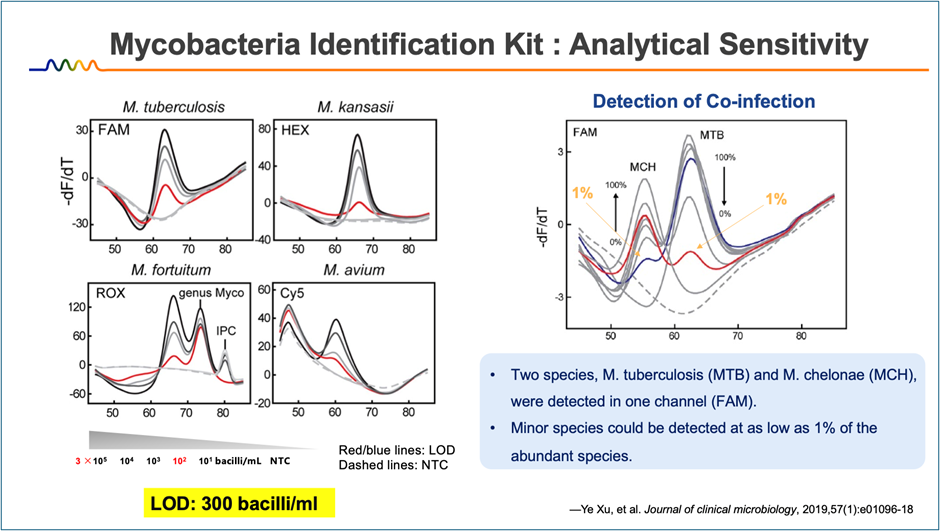
Rapid and Reliable Detection
Turnaround time: < 3 hours
Analytical sensitivity: platform-dependent
High specificity based on melting curve discrimination
Clinical Validation
The technology has been evaluated in international clinical settings, demonstrating:
High concordance with reference identification methods
Robust performance across diverse mycobacterial species
Performance characteristics may vary depending on study design and platform configuration.
Key Advantages
True High-Multiplex Capability
Simultaneous detection of multiple NTM species in a single reaction.
Faster Than Culture
Reduces time-to-identification from weeks to hours.
Clinically Actionable Results
Supports species-level differentiation relevant to treatment decisions.
Automated Interpretation
Reduces subjectivity and improves reproducibility.
Scalable Platform
Adaptable to both centralized and decentralized testing environments.
Applications
Clinical Microbiology Laboratories
Rapid identification of NTM from respiratory specimens.
Reference Laboratories
High-throughput species identification and confirmation.
Epidemiology & Surveillance
Species distribution and infection trend analysis.
Ordering Info:
Cat. No. | Description | Size |
20048 | Mycobacteria ldentification Kit | 48-reactions |
Mycobacteria ldentification Kit is a CE marked product, and for In Vitro Diagnostic Use.
Mycobacteria ldentification Kit is not FDA-cleared.